Ask for a reprint
email :
* Give your email
2018
ACL
|
Pierre Florian, Alexey Novikov, James Drewitt, Louis Hennet, Vincent Sarou-Kanian, Dominique Massiot, Henry Fischer, Daniel R.Neuville, 'Structure and Dynamics of High-Temperature Strontium Aluminosilicate Melts', Phys. Chem. Chem. Phys. 20 27865-27877 (2018) doi:10.1039/c8cp04908d
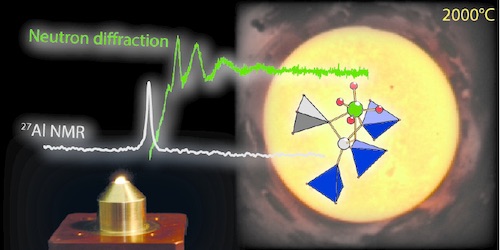
We report the study of high-temperature melts (1600°C-2300°C) and related glasses in the SrO-Al2O3-SiO2 phase diagram considering three series: (i) depolymerized ([SrO]/[Al2O3] = 3); (ii) fully polymerized ([SrO]/[Al2O3] = 1); and (iii) per-aluminous ([SrO]/[Al2O3] < 1). By considering results from high-temperature 27Al NMR and high temperature neutron diffraction, we demonstrate that the structure of the polymerized melts is controlled by a close-to-random distribution of Al and Si in the tetrahedral sites, while the depolymerized melts show smaller rings with a possible loss of Non-Bridging Oxygens on AlO4 units during cooling for high-silica compositions. Few five-fold coordinated VAl sites are present in all compositions except per-aluminous ones where high amounts of high-coordinated aluminium are found in the glasses and melts with complex temperature dependence. In the high-temperature melts Strontium has a coordination number of 8 or lower i.e. lower than in the corresponding glasses. The dynamics of the high-temperature melt were studied from 27Al NMR relaxation and compared to macroscopic shear viscosity data. These methods provide correlation times in close agreement. At very high temperatures, the NMR correlation times can be related to oxygen self-diffusion coefficient and we show a decrease of the latter with increasing Si/(Al+Si) ratio for polymerized melts with no compositional dependence for depolymerized ones. The dominant parameter controlling the temperature dependence of the aluminum environment of all melts is the distribution of Al-(OSi)p(OAl)(4-p) units.
|
|