Ask for a reprint
email :
* Give your email
2022
ACL
|
Verónica Poza-Nogueiras, Alicia Gomis-Berenguer, Marta Pazos, Angeles Sanroman, Conchi O.Ania, 'Exploring the use of carbon materials as cathodes in electrochemical advanced oxidation processes for the degradation of antibiotics', J. Env. Chem. Eng. 10 107506 (2022) doi:10.1016/j.jece.2022.107506
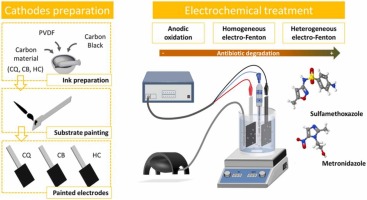
Three carbon materials from a varied origin, composition and porosity have been used as cost-effective cathodes in different electrochemical advanced oxidation processes (i.e. anodic oxidation (AO) and electro-Fenton (EF) in homogeneous and heterogeneous catalysis modalities) for the degradation of an antibiotic (e.g., metronidazole). In general, the efficiency towards the pollutant degradation increased as: AO < homogeneous EF < heterogeneous EF. The heterogeneous EF approach with iron pre-loaded in the carbon cathode outperformed the homogenous EF counterpart, despite a lower amount of iron. Reusability tests showed that iron-containing cathodes prepared from samples CQ (carbon obtained upon steam activation) and CB (carbon prepared by H3PO4 activation of biomass) were stable over at least three consecutive cycles. The best results were attained for cathode prepared with carbon CQ modified with iron, yielding over 97% removal of metronidazole after 150 min in all three reuses. Such better performance is related to the favorable adsorption of the pollutant on carbon CQ, thereby favoring the close contact between the generated oxidant species and the target antibiotic. The cathode based on carbon CQ was tested for the degradation of another recalcitrant antibiotic (sulfamethoxazole) in a real wastewater treatment plant effluent by means of a heterogeneous EF treatment, showing good degradation performance.
|
|